Treating brain tumors, like glioblastoma, is challenging. Current glioblastoma drugs, Rapamycin and Temozolomide (TMZ), generally only extend life for a matter of months, are not very effective in treating recurring tumors, and have significant clinical toxicity. A novel Plasma Discharge Tube (PDT) device has the potential to be more effective and less toxic. PDT may be used alone or may be used to enhance the efficacy of drugs. GW inventors previously developed a cold atmospheric plasma (CAP) therapy, which is in human trials for treating areas around surgically removed tumors to kill any cancer cells missed. PDT improves on the traditional CAP by enabling non-invasive treatment rather than requiring direct contact between the target tissue and the cold plasma.
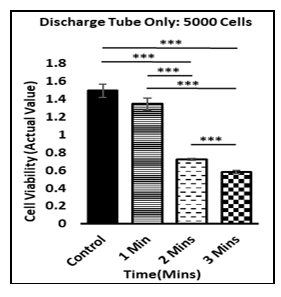
The operation of a plasma discharge tube is similar to a fluorescent light bulb. PDT generates an electromagnetic field (EMF) by plasma irradiation and then projects that field onto target cells. In vitro studies of cells exposed to EMF from CAP showed reactive oxygen species (ROS), DNA damage, and apoptosis result in selective death of cancer cells and synergy with TMZ.
Applications:
- Device to treat tumors, especially brain tumors like glioblastoma
Advantages:
- Non-invasive
- Synergistic with chemotherapy